
Arman Eshaghi
MS-PINPOINT study funded and due to start!
MS-PINPOINT study has been funded and is due to start looking at large-scale data by summer 2023. The summary of the study can be found below.
Background
In multiple sclerosis (MS), the immune system attacks the brain and spinal cord. This causes problems in how we move, feel or think. In the UK, 130,000 people live with MS, costing the NHS more than £2.9 billion a year.
MS types are based on when and how symptoms appear. Most cases (80%) begin as relapsing-remitting MS, with unpredictable attacks and remissions. After 10-20 years, about 50% of patients develop secondary progressive MS with gradually worsening symptoms without obvious attacks. In primary progressive MS, symptoms slowly worsen from the start and accumulate over many years.
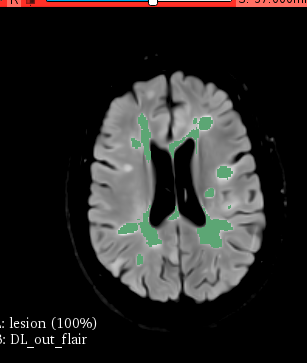
This classification does not reflect the underlying tissue changes (pathology), potential response to treatment or future disability. More than 15 treatments currently exist, each with different risks and strengths. Brain images, such as magnetic resonance imaging (MRI) and blood tests (serum neurofilament levels [sNfL]), help identify the underlying tissue changes. If types of MS were redefined based on the underlying tissue changes, doctors could recommend the most appropriate treatment with the fewest side effects.
Artificial intelligence (AI) uses computers for tasks that rely on human intelligence. . However, clinical trials have strict criteria for taking part. They typically exclude people in certain age ranges or with more than one lifelong condition. MS-PINPOINT will now develop new tools for real-world use from diverse patient populations to benefit anyone with MS.
Aim
MS-PINPOINT will identify patterns of MRI and serum neurofilament (where available) change that characterise specific types of MS. These types will predict how symptoms may change, when MS may worsen, and which drugs are most likely to help.
We will work with colleagues from several NHS Trusts (so far, 11 UK hospitals have expressed interest in participating) and two international clinical centres to:
1) Use MRI and, where available, sNfL to define MS types that reflect underlying abnormalities better than symptom-based types.
2) Build AI tools that measure and predict whether a treatment works at an individual level. This will make it easier to tell how new MS types predict treatment effects in NHS data.
3) Build AI tools based on objectives (1) and (2) that can learn from each other without sharing patients’ data across centres to optimise and refine the predictions. This ensures that we can include more patients from diverse backgrounds for truly personalised predictions.
I will measure how new MS types complement the existing MS types based on disability outcomes and calculate how my digital tools may save costs in future healthcare by recommending appropriate treatments earlier and delaying disability.
Benefits
The results by the end of 2027 will be research tools that predict whether a treatment works using widely available NHS data. It will give researchers information on the probability of treatment response and disease worsening. In the future, this could evolve into hospital software to help doctors recommend treatments, minimise disability accrual, reduce side effects and save money.
Patient and public involvement (PPI)
MS-PINPOINT was developed in consultation with a PPI steering committee. The committee will meet twice a year to continue to shape the research and advise on the acceptability of the future medical device software tool, interface, risks and benefits. The PPI committee will assist in designing a follow-on path for future use in clinical practice.
Dissemination
Please check our website, , for regular updates.
Arman Eshaghi
Arman is an NIHR Advanced Fellow who is leading the MS-PINPOINT project. He is an expert in machine learning applied to medical images and clinical outcomes in multiple sclerosis.
Leave a comment
Related Posts
PostwidgetCategories
